Your enquiry has been submitted
Your enquiry has been submitted
XLD Agar, Modified
Intended use
Recommended for selective isolation and enumeration of Salmonella Typhi and other Salmonella species. The composition and performance criteria of this medium are as per specifications laid down in ISO 6579-1:2017./ Amd: 2020, ISO 19250:2010(E) and APHA.
Composition**
ISO 6579-1 Specification - XLD Agar
| Ingredients | g/L |
|---|---|
| Yeast extract | 3.000 |
| L-Lysine hydrochloride | 5.000 |
| Lactose | 7.500 |
| Sucrose | 7.500 |
| Xylose | 3.750 |
| Sodium chloride (NaCl) | 5.000 |
| Sodium deoxycholate | 1.000 |
| Sodium thiosulphate | 6.800 |
| Iron (III) ammonium citrate | 0.800 |
| Phenol red | 0.080 |
| Agar | 9.00-18.00 |
Final pH (at 25°C): 7.4±0.2
M0311 - XLD Agar, Modified
| Ingredients | g/L |
|---|---|
| Yeast extract | 3.000 |
| L-Lysine hydrochloride | 5.000 |
| Lactose | 7.500 |
| Sucrose | 7.500 |
| Xylose | 3.750 |
| Sodium chloride | 5.000 |
| Sodium deoxycholate | 1.000 |
| Sodium thiosulphate | 6.800 |
| Ferric ammonium citrate# | 0.800 |
| Phenol red | 0.080 |
| Agar | 15.000 |
Final pH (at 25°C): 7.4±0.2
**Formula adjusted, standardized to suit performance parameters # - Equivalent to Iron (III) ammonium citrate
Directions
Suspend 55.43 grams in 1000 ml purified/distilled water. Heat with frequent agitation until the medium boils. DO NOT AUTOCLAVE OR OVERHEAT. Transfer immediately to a water bath at 50°C. After cooling, pour into sterile Petri plates. It is advisable not to prepare large volumes which will require prolonged heating. Note: Slight precipitation in the medium may occur, which is inheritant property of the medium, and does not affect the performance of the medium.
Principle And Interpretation
XLD Agar was formulated by Taylor (1-6) for the isolation and differentiation of enteric pathogens including Salmonella Typhi from other Salmonella species. XLD Agar, Modified (M0311) is recommended for selective isolation and enumeration of Salmonella species in accordance with ISO Committee, APHA (7-11). The incubation conditions has been revised as per the amendment 1, 2020 (8). The media formulation does not allow the overgrowth of other organisms over Salmonella and Shigella. The medium contains yeast extract, which provides nitrogen and vitamins required for growth. Though the sugars xylose, lactose and sucrose provide sources of fermentable carbohydrates, xylose is mainly incorporated into the medium since it is not fermented by Shigellae but practically by all enterics. This helps in the differentiation of Shigella species. Sodium chloride maintains the osmotic balance of the medium. Lysine is included to differentiate the Salmonella group from the non-pathogens. Salmonellae rapidly ferment xylose and exhaust the supply. Subsequently lysine is decarboxylated by the enzyme lysine decarboxylase to form amines with reversion to an alkaline pH that mimics the Shigella reaction. However, to prevent this reaction by lysine-positive coliforms, lactose and sucrose are added to produce acid in excess. Degradation of xylose, lactose and sucrose to acid causes phenol red indicator to change its colour to yellow. Bacteria that decarboxylate lysine to cadaverine can be recognized by the appearance of a red colouration around the colonies due to an increase in pH. These reactions can proceed simultaneously or successively, and this may cause the pH indicator to exhibit various shades of colour or it may change its colour from yellow to red on prolonged incubation. To add to the differentiating ability of the formulation, an H2S indicator system, consisting of sodium thiosulphate and ferric ammonium citrate, is included for the visualization of hydrogen sulphide produced, resulting in the formation of colonies with black centers. The non-pathogenic H2S producers do not decarboxylate lysine therefore, the acid reaction produced by them prevents the blackening of the colonies. XLD Agar is both selective and differential medium. It utilizes sodium deoxycholate as the selective agent and therefore it is inhibitory to gram-positive microorganisms.
Type of specimen
Food and meat samples. milk and milk products, animal feed, animal faeces, environmental samples, Water samples
Specimen Collection and Handling:
Processing: (7)
Pre-enrichment: Samples (25 grams in 225 ml) are pre-enriched in Buffered Peptone Water (M1494I)/(GM14941) and incubated at 34°C to 38°C for 18 h± 2 hours.
Selective enrichment: 0.1 ml of pre-enriched sample is inoculated in 10 ml RVS Broth (M1448I) or MSRV Agar (M1428) and incubated at 41.5 ± 1°C for 24 ± 3 hours and 1 ml of culture is inoculated in MKTTn broth (M1496I) and incubated at 36±2°C for 24 ± 3 hours.
Isolation: The culture thus obtained is then plated on XLD Agar, Modified (M0311) and incubated at 36° ± 2°C for 24 ± 3 hours. Simultaneously plating on second isolation agar is carried out.
Confirmation: Biochemical and serological tests are performed for confirmation.
Warning and Precautions
Read the label before opening the container. Wear protective gloves/protective clothing/eye protection/ face protection. Follow good microbiological lab practices while handling specimens and culture. Standard precautions as per established guidelines should be followed while handling specimens. Safety guidelines may be referred in individual safety data sheets.
Limitations
- Slight precipitation in the medium may occur, which is inheritant property of the medium,and does not affect the performance of the medium.
- XLD Agar is based on fermentation reaction and H2S production hence second medium should be selected so as to detect lactose positive and H2S negative strains.
- S.Paratyphi A, S.choleraesuis, S.pullorum and S.gallinarum may form red colonies without H2S, thus resembling Shigella species.
- Atypical Salmonella species which are lactose positive and/or H2S negative should be confirmed by biochemical and serological tests.
Performance and Evaluation
Performance of the medium is expected when used as per the direction on the label within the expiry period when stored at recommended temperature.
Quality Control
Appearance Light yellow to pink homogeneous free flowing powder
Gelling Firm, comparable with 1.5% Agar gel
Colour and Clarity of prepared medium Red coloured clear to slightly opalescent gel forms in Petri plates
Reaction Reaction of 5.54% w/v aqueous solution at 25°C. pH : 7.4±0.2
pH 7.20-7.60
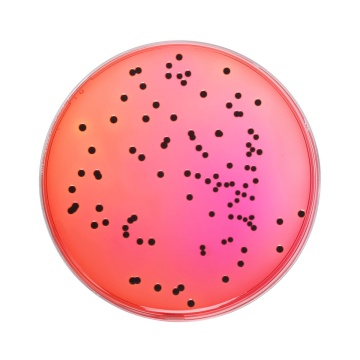
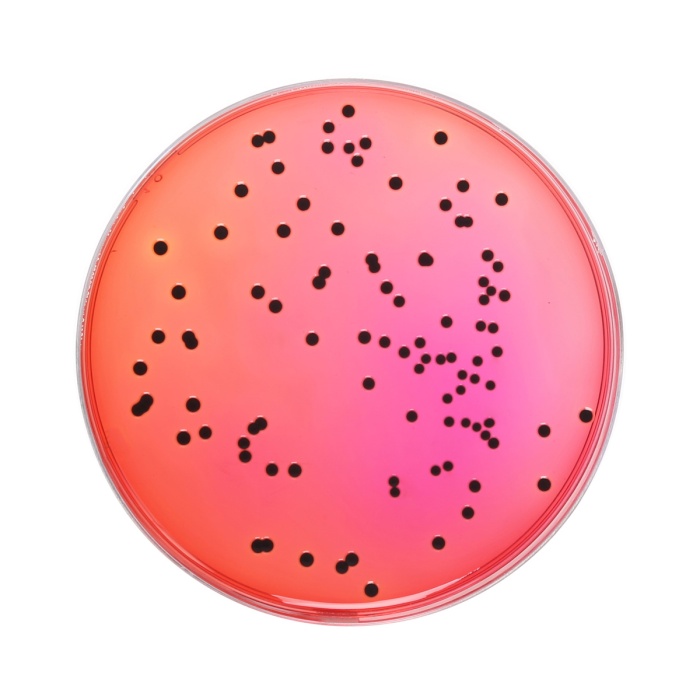
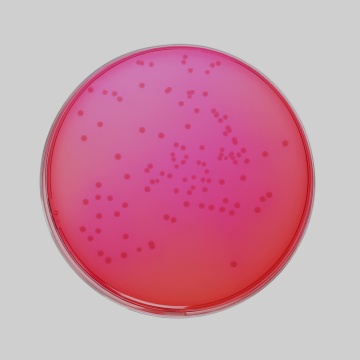
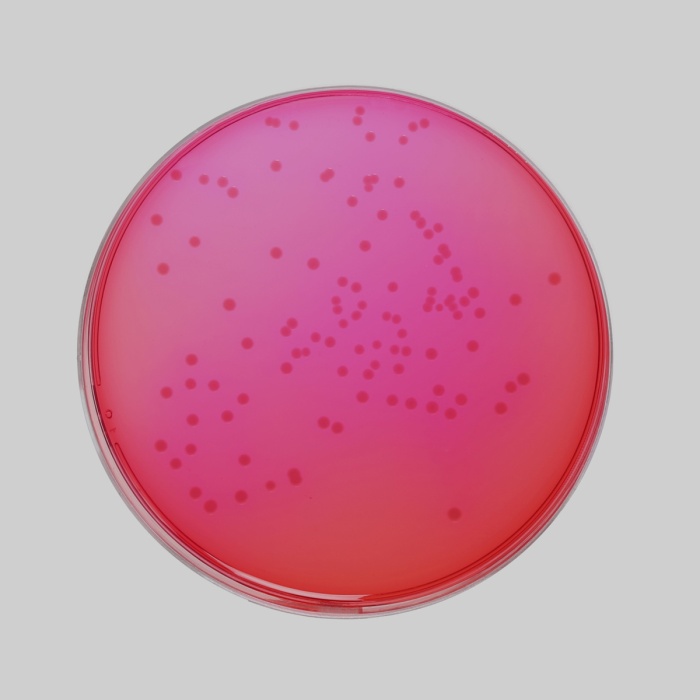
Cultural Response Cultural response was observed after an incubation at 34°C to 38°C for for 24 ± 3 hours. Recovery rate is considered as 100% for bacteria growth on Soyabean Casein Digest Agar.
| Organism | Inoculum (CFU) | Growth | Recovery | Colour of Colony |
|---|---|---|---|---|
| Productivity | ||||
| Salmonella Typhimurium ATCC 14028 (00031*) | 50-100 | good | >=50 % | red with black centres |
| Salmonella Enteritidis ATCC 13076 (00030*) | 50-100 | good | >=50 % | red with black centres |
| Selectivity | ||||
| Escherichia coli ATCC 8739 (00012*) | >=104 | growth or partial inhibition | yellow | |
| Escherichia coli ATCC 25922 (00013*) | >=104 | growth or partial inhibition | yellow | |
| Enterococcus faecalis ATCC 29212 (00087*) | >=104 | inhibited | 0 % | - |
| Enterococcus faecalis ATCC 19433 (00009*) | >=104 | inhibited | 0 % | - |
Key: (*) Corresponding WDCM numbers.
Please refer disclaimer Overleaf.
Storage and Shelf Life
Store between 10-30°C in a tightly closed container and the prepared medium at 20-30°C. Use before expiry date on the label. On opening, product should be properly stored dry, after tightly capping the bottle in order to prevent lump formation due to the hygroscopic nature of the product. Improper storage of the product may lead to lump formation. Store in dry ventilated area protected from extremes of temperature and sources of ignition Seal the container tightly after use.
Product performance is best if used within stated expiry period.
Disposal
User must ensure safe disposal by autoclaving and/or incineration of used or unusable preparations of this product. Follow established laboratory procedures in disposing of infectious materials and material that comes into contact with sample must be decontaminated and disposed of in accordance with current laboratory techniques (12,13).
Reference
- Taylor W. L., 1965, Am. J. Clin. Pathol., 44:471-475.
- Taylor W. L. and Harris B., 1965, Am. J. Clin. Pathol., 44:476.
- Taylor W. L. and Harris B., 1967, Am. J. Clin. Pathol., 48:350.
- Taylor W. L. and Schelhart B., 1967, Am. J. Clin. Pathol., 48:356.
- Taylor W. L. and Schelhart B., 1968, Am. J. Clin. Pathol., 16:1387.
- Taylor W. L. and Schelhart B., 1969, Appl. Microbiol., 18.393-395.
- Microbiobiology of the food chain- Horizontal method for the detection, enumeration and serotyping of Salmonella- Part I Detection of Salmonella. International Organization for Standardization (ISO), ISO/DIS 6579-1:2017.
- Microbiobiology of the food chain- Horizontal method for the detection, enumeration and serotyping of Salmonella- Part I Detection of Salmonella. International Organization for Standardization (ISO), ISO/DIS 6579-1:2017/ Amd 1:2020.
- Water Quality - Detection of Salmonella spp. International Organization for Standardization (ISO), ISO 19250:2010 (E).
- Salfinger Y., and Tortorello M.L., 2015, Compendium of Methods for the Microbiological Examination of Foods, 5th Ed., American Public Health Association, Washington, D.C.
- Lipps WC, Braun-Howland EB, Baxter TE, eds. Standard methods for the Examination of Water and Wastewater, 24th ed. Washington DC:APHA Press; 2023.
- Isenberg H. D., Kominos S., and Sigeal M., 1969, Appl Microbiol., 18, 656-659.
- Jorgensen, J.H., Pfaller, M.A., Carroll, K.C., Funke, G., Landry, M.L., Richter, S.S and Warnock., D.W. (2015) Manual of Clinical Microbiology, 11th Edition. Vol. 1.
| Product Name | XLD Agar, Modified |
|---|---|
| SKU | M031I |
| Product Type | Regular |
| Physical Form | Powder |
| Origin | Animal |
| Packaging type | HDPE |
| References | 1. Taylor W. L., 1965, Am. J. Clin. Pathol., 44:471-475. 2.Taylor W. L. and Harris B., 1965, Am. J. Clin. Pathol., 44:476. 3.Taylor W. L. and Harris B., 1967, Am. J. Clin. Pathol., 48:350. 4.Taylor W. L. and Schelhart B., 1967, Am. J. Clin. Pathol., 48:356. 5.Taylor W. L. and Schelhart B., 1968, Am. J. Clin. Pathol., 16:1387. 6.Taylor W. L. and Schelhart B., 1969, Appl. Microbiol., 18.393-395. 7.Chadwick P., Delisle G. H and Byer M., 1974, Can. J. Microbiol., 20, 1653-1664. 8.Salfinger Y., and Tortorello M.L. Fifth (Ed.), 2015, Compendium of Methods for the Microbiological Examination ofFoods, 5th Ed., American Public Health Association, Washington, D.C. 9.Baird R.B., Eaton A.D., and Rice E.W., (Eds.), 2015, Standard Methods for the Examination of Water andWastewater, 23rd ed., APHA, Washington, D.C. 10.Wehr H. M. and Frank J. H., 2004, Standard Methods for the Microbiological Examination of Dairy Products, 17th Ed.,APHA Inc., Washington, D.C. 11. Williams H., (Ed.), 2005, Official Methods of Analysis of the Association of Official Analytical Chemists, 19th Ed., AOAC,Washington, D.C.1 2.FDA Bacteriological Analytical Manual, 2005, 18th Ed., AOAC, Washington, D.C. 13.Dunn C. and Martin W. J., 1971, Appl. Microbiol., 22, 17-22.1 4.Rollender M. A., Beckford O., Belsky R. D and Kostroff B. 1969, Am. J. Clin. Pathol., 51, 284-286. 15.Taylor W. L. and Schelhart B., 1969, Appl. Micro. 18, 1387-1392. 16.MacCarthy M. D., 1966, N. Z. J. Med. Lab. Technol., 20, 127-131. 17.Isenberg H. D., Kominos S., and Sigeal M., 1969, Appl Microbiol., 18, 656-659.1 20.Isenberg, H.D. Clinical Microbiology Procedures Handbook. 2nd Edition.21. Jorgensen,J.H., Pfaller , M.A., Carroll, K.C., Funke, G., Landry, M.L., Richter, S.S and Warnock., D.W. (2015)Manual of Clinical Microbiology, 11th Edition. Vol. 1. |